Tuesday Poster Session
Category: Liver
P3839 - Outcomes of Liver Transplantation in CMV-seropositive Patients of South Asian Descent: A Retrospective Cohort Study
Tuesday, October 24, 2023
10:30 AM - 4:00 PM PT
Location: Exhibit Hall

Azhar Hussain, MD
SUNY Upstate Medical University, New York
Syracuse, New York
Presenting Author(s)
Abdul Wahab Dogar, MBBS, FCPS1, Azhar Hussain, MD2, Kaleem Ullah, MBBS, FCPS1, Shams Ud-Din, MBBS, FCPS1, Ameer Hamza, MBBS, FCPS1, Abdul Ghaffar, MBBS, FCPS1, Abu Hurairah, MD3, Ather Naeem, MBBS1, Muhammad Moseeb Ali Hashim, MBBS1
1Pir Abdul Qadir Shah Jeelani Institute of Medical Sciences, Gambat, Sindh, Pakistan; 2SUNY Upstate Medical University, New York, Syracuse, NY; 3AdventHealth, Orlando, FL
Introduction: Human cytomegalovirus (CMV) plays an important role in solid organ transplants, especially liver transplants. It increases the morbidity and mortality of liver transplant patients, so the testing of CMV in both donors and recipients is useful to predict liver transplant outcomes.
Methods: In this retrospective cohort study, 366 patients underwent liver transplantations during 2020-2022. Their medical reports were analyzed to see the impact of pretransplant CMV seropositivity on the outcomes of LDLT. CMV infection was diagnosed by serological and CMV PCR tests. Early allograft dysfunction (EAD), postoperative complications (biliary & vascular), and three-year survival were compared.
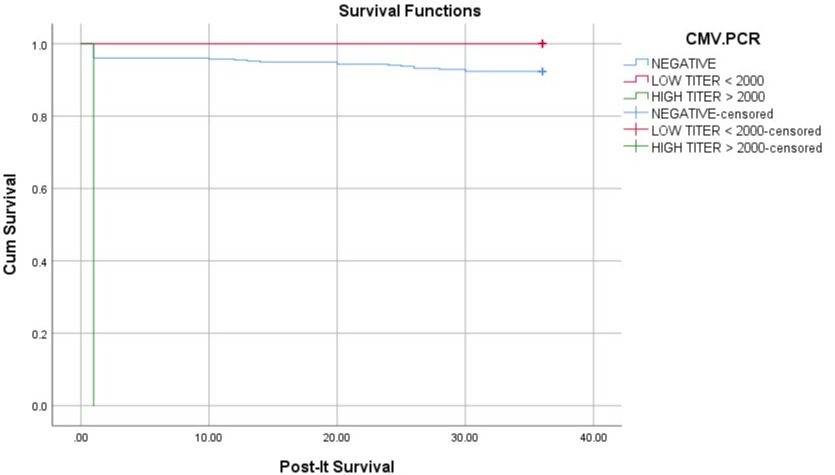
Results: In this study, 317 (86%) patients were male and 49 (14%) were female. In this study, 350 (95%) patients were pre-op CMV PCR-negative, 15 (4%) patients were pre-op CMV PCR-positive with low titer, and only 1 (0.2%) patient was pre-op CMV PCR-positive with high titer and was treated till resolution of CMV viremia with subsequent liver transplant. There was no statistically significant difference between the mean of the rest of the lab reports on the 1st and 7th post-op day, and liver transplant outcomes including ACR, EAD, and biliary & vascular complications (p >0.05). There was no significant difference in the survival time among the study groups on 3-year follow-up (log-rank test p=0.243).
Discussion: The purpose of this retrospective cohort study involving 366 LDLT adult recipients was to evaluate post-liver transplantation outcomes of pretransplant CMV-seropositivity. Out of 366 adult recipients, only 15 (4%) patients were having positive pre-transplant CMV PCR with low titer. 83 had positive post-op CMV PCR [low titer (< 1000 IU/mL)=79 & high titer (≥1000 IU/mL)=4] indicating post-op CMV reactivation or persistence of pre-transplant CMV viremia. Patients who had either pre-op or post-op positive CMV PCR were treated with valganciclovir at recommended doses which led to the resolution of CMV viremia in all those positive recipients uneventfully. No CMV prophylaxis was given in seronegative and aviremic recipients. There was no significant difference in the survival time among the study groups (log-rank test p=0.243).
We conclude that the positivity of CMV PCR in pre-transplant work-up with low titer viremia should not hinder the decision of pursuing LDLT, especially in emergent cases, as it does not lead to any significant adverse outcomes and comparable survival outcomes 3 years post-LDLT.
Disclosures:
Abdul Wahab Dogar, MBBS, FCPS1, Azhar Hussain, MD2, Kaleem Ullah, MBBS, FCPS1, Shams Ud-Din, MBBS, FCPS1, Ameer Hamza, MBBS, FCPS1, Abdul Ghaffar, MBBS, FCPS1, Abu Hurairah, MD3, Ather Naeem, MBBS1, Muhammad Moseeb Ali Hashim, MBBS1. P3839 - Outcomes of Liver Transplantation in CMV-seropositive Patients of South Asian Descent: A Retrospective Cohort Study, ACG 2023 Annual Scientific Meeting Abstracts. Vancouver, BC, Canada: American College of Gastroenterology.
1Pir Abdul Qadir Shah Jeelani Institute of Medical Sciences, Gambat, Sindh, Pakistan; 2SUNY Upstate Medical University, New York, Syracuse, NY; 3AdventHealth, Orlando, FL
Introduction: Human cytomegalovirus (CMV) plays an important role in solid organ transplants, especially liver transplants. It increases the morbidity and mortality of liver transplant patients, so the testing of CMV in both donors and recipients is useful to predict liver transplant outcomes.
Methods: In this retrospective cohort study, 366 patients underwent liver transplantations during 2020-2022. Their medical reports were analyzed to see the impact of pretransplant CMV seropositivity on the outcomes of LDLT. CMV infection was diagnosed by serological and CMV PCR tests. Early allograft dysfunction (EAD), postoperative complications (biliary & vascular), and three-year survival were compared.
Results: In this study, 317 (86%) patients were male and 49 (14%) were female. In this study, 350 (95%) patients were pre-op CMV PCR-negative, 15 (4%) patients were pre-op CMV PCR-positive with low titer, and only 1 (0.2%) patient was pre-op CMV PCR-positive with high titer and was treated till resolution of CMV viremia with subsequent liver transplant. There was no statistically significant difference between the mean of the rest of the lab reports on the 1st and 7th post-op day, and liver transplant outcomes including ACR, EAD, and biliary & vascular complications (p >0.05). There was no significant difference in the survival time among the study groups on 3-year follow-up (log-rank test p=0.243).
Discussion: The purpose of this retrospective cohort study involving 366 LDLT adult recipients was to evaluate post-liver transplantation outcomes of pretransplant CMV-seropositivity. Out of 366 adult recipients, only 15 (4%) patients were having positive pre-transplant CMV PCR with low titer. 83 had positive post-op CMV PCR [low titer (< 1000 IU/mL)=79 & high titer (≥1000 IU/mL)=4] indicating post-op CMV reactivation or persistence of pre-transplant CMV viremia. Patients who had either pre-op or post-op positive CMV PCR were treated with valganciclovir at recommended doses which led to the resolution of CMV viremia in all those positive recipients uneventfully. No CMV prophylaxis was given in seronegative and aviremic recipients. There was no significant difference in the survival time among the study groups (log-rank test p=0.243).
We conclude that the positivity of CMV PCR in pre-transplant work-up with low titer viremia should not hinder the decision of pursuing LDLT, especially in emergent cases, as it does not lead to any significant adverse outcomes and comparable survival outcomes 3 years post-LDLT.

Figure: Kaplan-Meier analysis showing comparable 3-year post-LDLT survival among different study groups (p= 0.243)
Disclosures:
Abdul Wahab Dogar indicated no relevant financial relationships.
Azhar Hussain indicated no relevant financial relationships.
Kaleem Ullah indicated no relevant financial relationships.
Shams Ud-Din indicated no relevant financial relationships.
Ameer Hamza indicated no relevant financial relationships.
Abdul Ghaffar indicated no relevant financial relationships.
Abu Hurairah indicated no relevant financial relationships.
Ather Naeem indicated no relevant financial relationships.
Muhammad Moseeb Ali Hashim indicated no relevant financial relationships.
Abdul Wahab Dogar, MBBS, FCPS1, Azhar Hussain, MD2, Kaleem Ullah, MBBS, FCPS1, Shams Ud-Din, MBBS, FCPS1, Ameer Hamza, MBBS, FCPS1, Abdul Ghaffar, MBBS, FCPS1, Abu Hurairah, MD3, Ather Naeem, MBBS1, Muhammad Moseeb Ali Hashim, MBBS1. P3839 - Outcomes of Liver Transplantation in CMV-seropositive Patients of South Asian Descent: A Retrospective Cohort Study, ACG 2023 Annual Scientific Meeting Abstracts. Vancouver, BC, Canada: American College of Gastroenterology.
